Post written by Douglas G. Adler, MD, FACG, AGAF, FASGE, from the Center for Advanced Therapeutic Endoscopy, Centura Health, Denver, Colorado, USA, and incoming GIE Editor-in-Chief.

We analyzed the U.S. Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database for all reports of patient-related adverse events and device failures associated with the 3 mechanical lithotripters most commonly used during ERCP in the United States.
All these devices are in widespread use in the United States, but they have very different mechanical properties, are made of different materials, and operate in some different ways. There were essentially no comparative data to guide end users on choosing between devices, no information on which devices experienced which issues, and so forth.
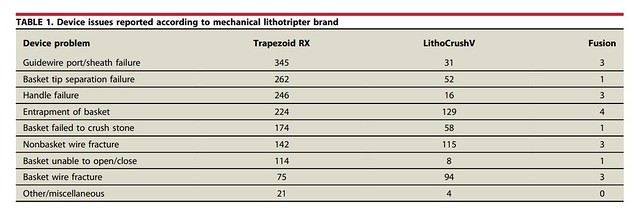
The number of device failures and patient-related adverse events were large (2129 and 205, respectively). Most device malfunctions were because of guidewire port or sheath failure (n = 379, 17.8%), basket entrapment (n = 357, 16.8%), and basket tip separation failure (n = 315, 14.8%).
The most commonly reported adverse events to patients were an additional procedure to remove an entrapped basket (170/205, 82.9%), bleeding (15/205, 7.3%), and bowel perforation (5/205, 2.4%). In the study, we break this down further by device type.
Understanding device malfunctions and patient-related adverse events regarding the use of ERCP mechanical lithotripters is crucial to their safe operation. In addition, understanding the mechanisms by which devices can fail can help inform device selection and operation and may reduce risk of device failure and patient-related adverse events.
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
