 Post written by Eelco C. Brand from the Department of Gastroenterology and Hepatology, University Medical Center Utrecht, Utrecht, the Netherlands.
Post written by Eelco C. Brand from the Department of Gastroenterology and Hepatology, University Medical Center Utrecht, Utrecht, the Netherlands.
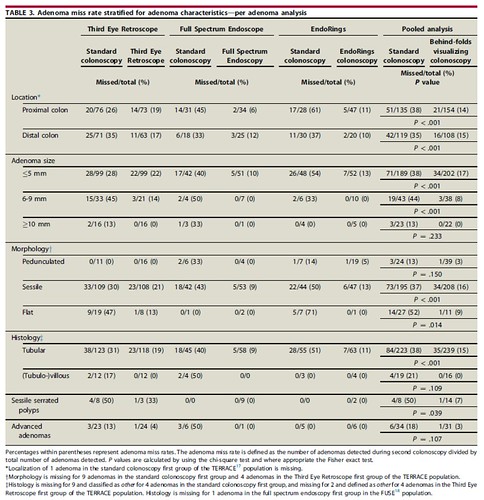
The efficacy of screening and surveillance colonoscopy is reduced by the occurrence of interval-/post-colonoscopy colorectal cancer. One of the factors associated with the occurrence of interval-/post-colonoscopy colorectal cancer is the fact that polyps and adenomas are missed during colonoscopy. Colorectal adenomas can be missed due to their localization in inner curves, behind colonic folds or in the proximal colon, or due to flat morphology. As a consequence, improvement of colonoscopy technologies has focused on designing colonoscopes or devices that are able to visualize the mucosa behind colonic folds. Three behind-folds visualizing colonoscopy technologies, ie, the Third Eye Retroscope, Full Spectrum Endoscope (FUSE), and EndoRings device, have recently been shown in randomized back-to-back colonoscopy studies to reduce overall adenoma miss rates. However, the question remains, what types of colorectal adenomas and in what type of patients adenomas are less frequently missed with these behind-folds visualizing technologies. Although a decrease in the overall adenoma miss rate is encouraging for the improvement of colonoscopy quality, emerging evidence suggests that certain subgroups of adenomas, eg, with villous features, ≥10mm, or with high-grade dysplasia (ie, advanced adenomas), and sessile serrated lesions are the most clinically relevant precancerous lesions. Furthermore, particularly flat and proximally located lesions are typically missed, and thus physicians could benefit from technologies that aid in the detection of these lesions. Analyzing which subgroups of colorectal lesions and in what type of patients adenomas are less frequently missed by the use of behind-folds visualizing colonoscopy technologies can therefore aid the assessment of the clinical benefit of these behind-folds visualizing colonoscopy technologies.

Figure 1. Three behind-folds visualizing colonoscopy technologies. A, the Third Eye Retroscope is advanced through the biopsy channel and provides a 180o retroflexed view on a second screen during withdrawal. B, The Full Spectrum Endoscope has 3 imagers and light emitting diode groups positioned at the front and at both sides of the distal tip of the colonoscope, providing a 330o view on 3 contiguous monitors. C, The EndoRings device is mounted on the distal tip of the colonoscope and consists of silicon circular wings that flatten the colonic folds during withdrawal.
In our pooled analyses of 3 randomized back-to-back tandem colonoscopy trials, we found that the use of behind-folds visualizing colonoscopy technologies reduces miss rates for 1 to 9 mm adenomas in all segments of the colon and for sessile serrated polyps, whereas no advantage was found for ≥10 mm and advanced adenomas, compared to standard forward-viewing colonoscopy. The adenoma miss rates were statistically significantly lower for all patients, regardless of sex, age >50 years, and the colonoscopy indication.
It is tempting to speculate that the reductions in adenoma miss rates will influence the occurrence and mortality of interval-colorectal cancer, but studies with long-term follow-up will be needed to assess the influence of behind-folds visualizing colonoscopy on this clinically important endpoint. As the colonoscopies in the 3 randomized trials were performed by experienced endoscopists, it would be of interest to analyze what the effects of these technologies are for physicians with low-baseline adenoma detection rates. Apart from that, cost-effectiveness analyses taking, for example, reusability of the device into account are awaited. Finally, possible disadvantages of the device, eg, increased diameter of the tip of the colonoscope with the EndoRings device, should be balanced against the advantages, eg, decrease in adenoma miss rate, when deciding on the use of a specific colonoscopy technology in daily practice.
Find the article abstract here.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
