 Douglas G. Adler, MD, FACG, AGAF, FASGE, from the Division of Gastroenterology and Hepatology at the University of Utah Hospital in Salt Lake City, Utah, USA discusses this Original Article “EUS-guided drainage of peripancreatic fluid collections and necrosis by using a novel lumen-apposing stent: a large retrospective, multicenter U.S. experience (with videos).”
Douglas G. Adler, MD, FACG, AGAF, FASGE, from the Division of Gastroenterology and Hepatology at the University of Utah Hospital in Salt Lake City, Utah, USA discusses this Original Article “EUS-guided drainage of peripancreatic fluid collections and necrosis by using a novel lumen-apposing stent: a large retrospective, multicenter U.S. experience (with videos).”
My co-lead author Ali Siddiqui MD and I both had significant experience with the new Lumen Apposing Metal Stent (LAMS) that was available in the United States, but we were interested in pooling the data from our centers with that from other centers to create a larger dataset to analyze outcomes when using these devices to drain pancreatic fluid collections (PFCs). Thus, the objective of this large multicenter study was to evaluate the overall clinical outcomes, success rate, and adverse events of the LAMS for EUS-guided transmural drainage of patients with symptomatic PFCs in the United States, as much of the early data on this device (and other similar devices) came from Europe and other locations. LAMSs are still a relatively new and novel technology that many have heard about but that, in reality, few have used. We thought that another small series of patients undergoing PFC drainage with LAMS would be less valuable than a large, multicenter study. 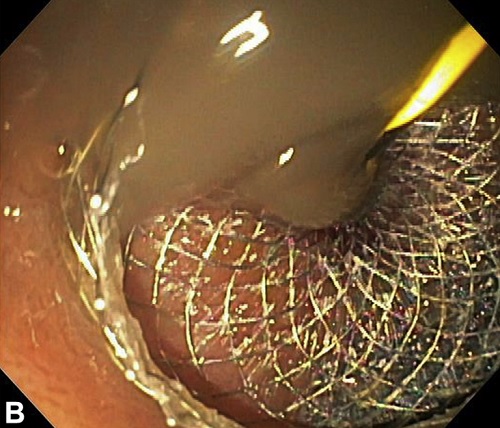
Figure 3B, Endoscopic image of the LAMS after deployment across the cystogastrostomy tract.
We studied patients with pancreatic pseudocysts and walled-off pancreatic necrosis. The average size of the PFC drained in our study was fairly large at 11.8 cm. LAMSs were successfully placed in 80 patients (97.5%). Twelve patients had pseudocysts (PP) and 68 patients had walled off necrosis (WON). The median stent in-dwell time was 2 months. Endoscopic debridement performed though the LAMS was performed in 54 patients with WON. The patency of the stent was maintained in 98.7% of the patients (77/78). There was spontaneous dislodgment of 2 LAMSs.
Successful endoscopic therapy by using the LAMS was successful in 12 of 12 patients (100%) with PP compared with 60 of 68 patients (88.2%) with WON, illustrating that patients with WON are sicker, require more involved treatment, and will have an overall lower success rate when compared to PP patients. Our overall success rate was low, with only 1 PFC recurrence during the 3-month follow-up period. Procedure-related adverse events occurred in 8 patients (9.8%), and included stent maldeployment in 2 patients,self-limited bleeding in 6 patients, and 1 patient with stent maldeployment developed gastric perforation which had to be repaired surgically.
Overall the study showed that LAMS are highly effective for the treatment of both PP and WON, but were overall more successful in patients with PP given the lack of need for endoscopic debridement. Furthermore, the safety profile of using these devices was within acceptable limits, recognizing that many of these patients are fairly ill to begin with. Our use of these devices has since continued to expand since the study was completed.
Read the abstract online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
