Authors Dr. Nicholas Burgess and Dr. Michael Bourke from Westmead Hospital and the University of Sydney in Sydney, NSW, Australia discuss their Thinking Outside the Box article “Sessile serrated adenomas/polyps with cytologic dysplasia: a triple threat for interval cancer” from the August issue.
Sessile serrated adenoma/polyps (SSA/P) are now recognized to play an important role in the pathogenesis of colorectal cancer accounting for 20-30% of sporadic cases. SSA/P have also been shown to be strongly associated with cancer that occurs in the surveillance period after complete colonoscopy, termed “interval cancer.” There are several epidemiologic, endoscopic, and pathologic aspects of SSA/P with cytological dysplasia (SSA/P-D) which reinforce their status as high risk interval cancer precursors.
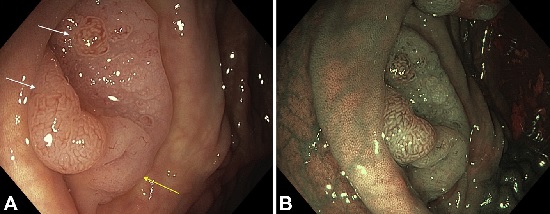 Figure 3. A, With a more contemporary colonoscope (Olympus 190 HD; Olympus, Tokyo, Japan), a 30-mm 0-IIa þ Is lesion was identified in the proximal transverse colon. This lesion contained 2 distinct nodular regions with “adenomatous” (Kudo III) pit patterns (white arrows). The majority of the lesion is flat (0-IIa morphology) and has typical features of a sessile serrated adenoma, with a pale surface and lacy capillaries, particularly at the 6 o’clock area (yellow arrow). An endoscopist who fails to carefully interrogate the mucosal field within which these “adenomatous nodules” are located easily could fail to recognize the true extent of the lesion and thus incompletely resect it. B, With narrow-band imaging, the 2 dysplastic nodules are well demarcated and more distinct from the surrounding serrated lesion and normal background mucosa.
Figure 3. A, With a more contemporary colonoscope (Olympus 190 HD; Olympus, Tokyo, Japan), a 30-mm 0-IIa þ Is lesion was identified in the proximal transverse colon. This lesion contained 2 distinct nodular regions with “adenomatous” (Kudo III) pit patterns (white arrows). The majority of the lesion is flat (0-IIa morphology) and has typical features of a sessile serrated adenoma, with a pale surface and lacy capillaries, particularly at the 6 o’clock area (yellow arrow). An endoscopist who fails to carefully interrogate the mucosal field within which these “adenomatous nodules” are located easily could fail to recognize the true extent of the lesion and thus incompletely resect it. B, With narrow-band imaging, the 2 dysplastic nodules are well demarcated and more distinct from the surrounding serrated lesion and normal background mucosa.
In contrast to conventional adenomas, SSA/Ps typically have little if any cytological dysplasia and are characterized by their hyperplastic and serrated architectural changes. It is likely that the transition to cancer in SSA/Ps occurs via the development of cytological dysplasia often resulting in the dysplastic area displaying a histological appearance indistinguishable from conventional adenoma. There is currently little data on SSA/Ps with cytological dysplasia (SSA/P-D), however based on the knowledge we do have, there are several aspects of SSA/P-D that mean they are likely to pose a significant risk for interval cancer. Firstly, SSA/P and SSA/P-D evade detection. SSA/P are difficult to see and several studies have shown they are easily overlooked. Endoscopically SSA/P-D is often associated with a change in surface morphology or a nodule within what appears to be a typical SSA/P reflecting the underlying histological alterations. In some cases, the dysplasia can be so dominant that the lesion visually mimics conventional adenoma. Second, SSA/P-D are likely to be at higher risk of incomplete resection. Studies have shown that residual polyp is common after snare resection of SSA/Ps.
In addition to this, SSA/P-D can mimic conventional adenoma which may increase the risk of incomplete resection, as the endoscopist may only “see” and remove the dysplastic component resembling adenoma, leaving the unrecognized surrounding non-dysplastic component. Pathologists may likewise fail to recognize SSA/P-D if they disregard or overlook serrated histology in what appears to be a conventional adenoma, or if they receive only the dysplastic nodule in an incompletely resected specimen. Thirdly, SSA/P-D may be a rapidly progressive lesion. The molecular changes that are often found in dysplastic lesions are similar to those that occur in Lynch syndrome, which is known for its aggressive nature, and there are several case reports of rapid progression of serrated lesions. These features of rapid progression, evasion of detection, and foiled resection constitute the three “threats” that SSA/P-D poses for the development of interval cancer. It is crucial that both the endoscopist and the pathologist are aware of the concept of SSA/P-D and its peculiar characteristics of imperceptibility and mimicry. Refocused attention on awareness, recognition and resection may help in addressing interval cancer rates.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
