Post written by Hirotoshi Ishiwatari, MD, PhD, from the Department of Pancreatobiliary Medicine, Shizuoka Cancer Center, Shizuoka, Japan.

The study aimed to investigate the optimal techniques for EUS-guided tissue acquisition (EUS-TA) to achieve successful comprehensive genomic profiling (CGP) testing using commercially available assays.
Matched therapy based on commercially available CGP tests plays a crucial role in improving patients’ prognosis. Particularly in pancreatic cancer, therapeutic options are limited, and matched therapy may serve as an important treatment strategy for select patients. CGP testing requires a larger amount of tissue compared with conventional pathological diagnosis.
Recently, FoundationOne CDx (F1CDx; Foundation Medicine, Inc, Cambridge, Mass, USA) became commercially available and has been widely adopted worldwide. However, the optimal EUS-TA technique to obtain sufficient tissue for F1CDx remained unclear. Addressing this unmet need was the primary motivation for our study.
In our previous retrospective study,1 we proposed 3 optimal approaches for obtaining adequate samples for successful F1CDx testing:
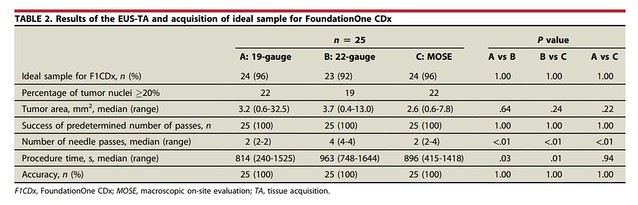
- Two passes with a 19-gauge (G) Franseen needle.
- Four passes with a 22-G Franseen needle.
- Macroscopic on-site evaluation (MOSE) using a 22-G Franseen needle until the macroscopic visible core length reached ≥40 mm.
The primary end point was the rate of acquisition of ideal samples meeting the F1CDx quantity criteria. Participants were randomized to 1 of the 3 methods, and each group’s success rate was compared with that of a historical control.
All 3 methods achieved success rates exceeding 90%, which were significantly higher than that of the historical control group. However, the 4-pass group required a longer procedure time and experienced 1 case of moderate abdominal bleeding. Based on these findings, we recommend 2 passes with a 19-G needle or MOSE using a 22-G needle as optimal EUS-TA strategies for successful F1CDx testing.
Our study also highlighted the importance of a combined embedding method when preparing formalin-fixed paraffin-embedded (FFPE) blocks. In this approach, a single FFPE block was created from all needle passes performed during the procedure. This technique increases the total amount of tissue included in the FFPE block submitted to Foundation Medicine, Inc.
The combined embedding method appears to play an essential role in achieving successful F1CDx testing. Therefore, when this preparation technique is combined with 2 passes using a 19-G needle or MOSE using a 22-G needle, the likelihood of successful CGP testing may be further enhanced. This strategy may facilitate the timely implementation of precision therapies, particularly when first-line treatment fails.
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
- Ishikawa K, Ishiwatari H, Sasaki K, et al. Optimization of endoscopic ultrasound-guided tissue sample acquisition for commercially available comprehensive genome profiling. J Gastroenterol Hepatol 2023;38:1794-1801. ↩︎
