Post written by Jae Hee Cho, MD, PhD, and Seungmin Bang, MD, PhD, from the Department of Internal Medicine, Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.

A double bare metal stent (DBS) is a newly developed metal stent that is woven into a cross and hook structure with braided nitinol, and it has a unique double-layered structure that crosses the 2 stents.
Characteristically, as the diameter of DBS cells (1-1.5 mm) is smaller than those of other metal stents (4 mm), the double-layered metal mesh efficiently prevents tumor ingrowth.
Our group previously reported that the stent patency of the DBS was longer than that of an uncovered single-layer metal stent. From different perspectives of the covering membrane of self-expandable metal stents (SEMSs), this randomized trial aimed to compare the clinical outcomes between the uncovered DBS (UCDBS) and the partially covered DBS (PCDBS) in patients with malignant biliary obstruction.

Of the 258 prospectively enrolled patients, 130 patients were randomly assigned to the PCDBS group and 128 to the UCDBS group. The clinical success rate was similar in the groups (96.9% vs 96.1%). The mean duration of stent patency of PCDBSs (421.2 days; 95% confidence interval [CI], 346.7-495.7) was longer than that of UCDBSs (377.4 days; 95% CI, 299.7-455.0), although total stent dysfunction and stent dysfunction within 6 months were not different between the groups.
In terms of adverse events, the rate of in-stent tumor ingrowth was significantly higher in the UCDBS group than in the PCDBS group (24.4% vs 13.4%). However, the incidence of other adverse events—including pancreatitis, cholangitis, cholecystitis, and bleeding—was similar between the 2 groups.
In addition, we evaluated the risk factors affecting stent patency and overall survival (OS) in this study. Although the chemotherapy regimen was not uniform, stent patency and OS were prolonged in patients who underwent chemotherapy compared with those in the supportive care group. This suggests that controlling tumor burden with cancer treatment could prolong stent patency and decrease the incidence of in-stent tumor ingrowth.
However, multivariate analysis using the Cox proportional hazard model indicated that chemotherapy was not significant (hazard ratio [HR], 1.605; 95% CI, .939-2.744; P = .84) for stent patency but significant for OS (HR, .572; 95% CI, .409-.799; P = .001).
In conclusion, we demonstrated that the use of PCDBSs does not alter stent patency rates at 6 months or mortality when compared with UCDBSs in patients with distal malignant biliary obstruction, although PCDBSs resulted in decreased stent ingrowth.
Yet, it was confirmed that the mean stent patency of UCDBSs and PCDBSs was longer than that of conventional uncovered single-layer SEMSs. Further studies are needed to clarify the optimal role of DBSs and to identify the patient population who may most benefit from DBSs as novel devices.
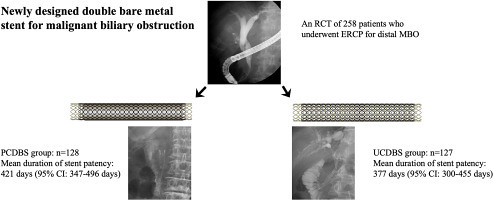
Graphical Abstract
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
