Post written by Antonio Facciorusso, MD, PhD, from the Endoscopy Unit, University of Foggia, Foggia, Italy.

EUS-guided tissue acquisition (EUS-TA) for cytology through fine-needle aspiration (FNA) or fine-needle biopsy (FNB) using specialized core needles has become a central technique in the assessment of pancreatic masses. The most important pitfall associated with this procedure is a false-negative diagnosis that has the potential to delay patient care and negatively impact patient outcomes. Among the variables that may potentially impact diagnostic characteristics of EUS-TA, the 2variables that play a pivotal role are the type of needle (fine-needle aspiration [FNA] and fine-needle biopsy [FNB]) and needle gauge (19-gauge vs 22-gauge vs 25-gauge). In particular, obtaining histologic specimens or core biopsies using EUS-FNB has generated a great deal of interest in the field of EUS-TA.
Hence, we performed pairwise and network meta-analysis comparing the diagnostic accuracy of EUS-FNA (22-gauge, 25-gauge, 19-gauge) and EUS-FNB (22-gauge, 25-gauge) needles for pancreatic masses. Grading of Recommendations Assessment, Development and Evaluation (GRADE) criteria for network meta-analysis were used to appraise quality of evidence.
There are currently limited data on the comparative diagnostic performance of different EUS-TA techniques, based on needle design and gauge, for pancreatic masses. Additionally, there is no systematic assessment of the quality of evidence, which can inform clinical guidelines.
As network meta-analysis involves the simultaneous analysis of direct evidence (from RCTs directly comparing diagnostic modalities of interest) and indirect evidence (from RCTs comparing diagnostic modalities of interest with a common comparator), such a systematic and comparative synthesis of the entire body of evidence, with critical appraisal of the quality of evidence can directly and optimally inform clinical practice guidelines.
We observed that there was no significant difference in diagnostic accuracy between different EUS-TA approaches for sampling pancreatic masses, based on low-quality evidence. In particular, there was no difference in the diagnostic accuracy between FNA versus FNB needles and between 22-gauge versus 25-gauge needles. Similarly, we found no significant difference between needle types and gauges for adequacy of samples, histological core procurement rate and number of needle passes. In this regard, it should be noted that direct comparisons based on single head-to-head trials showed a significant benefit with some FNB needles (25-gauge and Fork-Tip FNB) with respect to standard 25-gauge FNA in terms of sample adequacy and histological core procurement; however, given the paucity of such comparative studies, these findings did not impact significantly on network meta-analysis thus requiring a particular caution in interpreting these results.
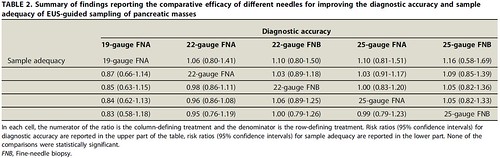
Our results suggest that EUS-FNA would suffice for most cases in routine clinical practice (patients with pancreatic adenocarcinoma) and add credence to the recently published European guidelines that equally recommend FNA and FNB for routine sampling of solid masses.
Our study was unable to examine the role of EUS-FNB versus EUS-FNA for conditions that require assessment of tissue architecture such as suspected autoimmune pancreatitis.
The promising results in tissue procurement and diagnostic performance observed with newer FNB needles such as the Franseen and Fork-tip needle need to be confirmed in further comparative RCTs, and the very limited number of available studies suitable to be included in our network meta-analysis did not allow us to draw definitive conclusions in this regard.
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
