 Post written by Cord Langner, MD, and Magdalena M. Gilg, MD, from the Institute of Pathology, Medical University Graz, Graz, Austria.
Post written by Cord Langner, MD, and Magdalena M. Gilg, MD, from the Institute of Pathology, Medical University Graz, Graz, Austria.
Secondary tumors of the gastrointestinal tract (GIT) are a rare finding with a dismal prognosis because they are only found with advanced cancers. This study aimed to investigate clinical, endoscopic, and pathological features of secondary tumors diagnosed during gastroscopy or colonoscopy.
Most studies on secondary tumors of the GIT include only small numbers of patients because they are usually case reports or retrospective analyses from single endoscopy units that include limited information on underlying primary tumors as well as clinical and endoscopic presentation. For a larger survey, a pathology-based retrospective observational study was designed to create the biggest cohort available so far, including all secondary tumors diagnosed at one pathology department.
Secondary tumors affected the GIT either by direct invasion (primarily to duodenum and rectum) or by lymphatic and/or hematogeneous spread (primarily to stomach). Malignant melanoma, breast cancer and pancreatic cancer were the most common underlying primaries.
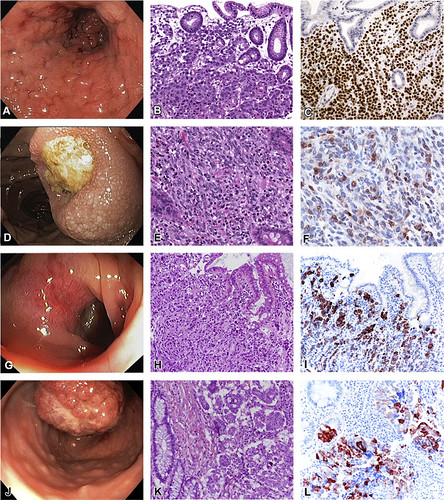
Figure 1. Examples of secondary tumors of the GI tract. A-C, Lobular breast cancer metastatic to the stomach. A, Intramural process (linitis plastica-like pattern) mimicking a diffuse gastric primary. B, Expansion of the stroma by atypical discohesive cells (H&E, orig. mag. ×100). C, Strong nuclear expression of estrogen receptor (orig. mag. ×100); the tumor cells also expressed keratin 7 and mammaglobin. D-F, Malignant melanoma metastatic to the duodenum. D, Submucosal process with volcano-like ulceration. E, Expansion of the stroma by cohesive atypical epithelioid and spindle cells (H&E, orig. mag. ×100). F, Immunoreactivity for melan-A (orig. mag. ×200); the tumor cells also expressed S100 protein and HMB-45. G-I, Gastric cancer infiltrating the transverse colon (direct infiltration). G, Submucosal (intramural) process. H, Diffuse infiltration of the stroma by atypical cells, growing as single cells or in small nests (H&E, orig. mag. ×100). I, Cytoplasmic expression of keratin 7; the tumor cells also expressed MUC5AC and MUC6 and were negative for SATB2. J-L, Ovarian cancer metastatic to the sigmoid colon. J, Polypoid mass mimicking a primary colonic neoplasm. K, Micropapillary tumor growth within unlined clear spaces (H&E, orig. mag. ×150). L, Cytoplasmic expression of keratin 7; the tumor cells also expressed CA125 and WT-1 and were negative for keratin 20 and SATB2.
Mean-time intervals between diagnoses of primary and secondary tumors are highly variable, with intervals less than 1 month for pancreatic cancer and up to 67 months for renal cell carcinomas.
It is of note that, about 1/4 of corresponding primary tumors were unknown at the time of endoscopy, that is, the secondary tumors were the first manifestation of the disease.
Notably, nearly every second lesion was diagnosed endoscopically as a primary tumor, in some cases even as a benign lesion. This can be explained by low incidence as well as uncharacteristic clinical and endoscopic appearance. Biopsy diagnosis is crucial to avoid misclassification.
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
