 Post written by Antti Siiki, MD, PhD, from the Department of Gastroenterology and Alimentary Tract Surgery, Tampere University Hospital, Tampere, Finland.
Post written by Antti Siiki, MD, PhD, from the Department of Gastroenterology and Alimentary Tract Surgery, Tampere University Hospital, Tampere, Finland.
Our group (Siiki, Rinta-Kiikka, Sand, and Laukkarinen) reported the first long-term results of endoscopically inserted biodegradable stents in human bile duct. The novel stents were used in 2 indications in ERCP with a standard duodenoscope: benign biliary strictures and post-cholecystectomy bile leaks. The main advantage of biodegradable stents is that second endoscopy for stent removal is not necessary. Secondly, the large stent diameter and expansion force may be beneficial compared to conventional stents in both indications. We feel very privileged to be among the pioneers to try and gather experience with the novel stents in ERCP in patients. Until now, there has been no endoscopic insertion device for biodegradable stents compatible with ERCP and duodenoscopes. While there were few previous results to compare with, the patient safety and stent biocompatibility were the main concerns to be critically evaluated.
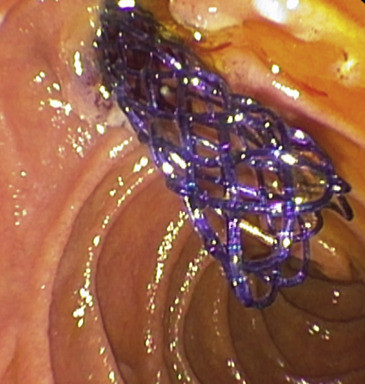
Figure 1. A braided 10-mm bore (60-mm length) self-expandable biodegradable poly-dioxanone biliary stent with golden radiopaque markers at both ends in situ as used in the study (Ella CS, Hradec Králové, Czech Republic). The stent was inserted across the papilla for treatment of postcholecystectomy leak of the cystic duct.
The biocompatibility and the first results with up to 2-years of follow-up in patients seem to be promising. We should continue with larger studies on biodegradable stents in ERCP. We believe that the full potential of biodegradable materials and stent design is still to be discovered. Improved endoscopic insertion devices, stent degradation profile, expansion force, and clinical efficacy in stricture management as well as the option for controlled drug release are some of the continuously developing issues that are eagerly looked forward to.
It is extremely interesting to see where the development of biodegradable stents and materials leads in the management of biliary diseases in ERCP.
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
