 Rungsun Rerknimitr, MD, Director of GI Endoscopy Excellence Center at the King Chulalongkorn Memorial Hospital in Bangkok, Thailand and President of the Thai Association of Gastrointestinal Endoscopy (TAGE) 2014-2015, discusses this Original Article “A combination of clinical risk stratification and fecal immunochemical test results to prioritize colonoscopy screening in asymptomatic participants.”
Rungsun Rerknimitr, MD, Director of GI Endoscopy Excellence Center at the King Chulalongkorn Memorial Hospital in Bangkok, Thailand and President of the Thai Association of Gastrointestinal Endoscopy (TAGE) 2014-2015, discusses this Original Article “A combination of clinical risk stratification and fecal immunochemical test results to prioritize colonoscopy screening in asymptomatic participants.”
The incidence of colorectal cancer (CRC) has rapidly increased in many Asian countries. Although colonoscopy is considered the most accurate test for the early detection and prevention of CRC, colonoscopy requires investment in expensive resources, including an endoscopy unit. In addition, many countries in Asia including Thailand have few well-trained endoscopists, which is incompatible with the colonoscopy screening volume. Hence, using colonoscopy as a primary screening method may not be feasible. We therefore proposed that a combination of the fecal immunochemical test (FIT) and clinical risk score to develop colon cancer would be helpful in selecting and prioritizing asymptomatic subjects for colonoscopy.
Based on an asymptomatic Asian population study, advanced age, family history of CRC, male gender, and smoking were identified as significant risk factors for risk score calculation. In our study subjects were classified as moderate-risk and high-risk groups. Nine-hundred and forty eight asymptomatic subjects underwent a complete colonoscopy. With the combination of clinical risk score and FIT results, subjects were classified into 4 groups; 84 subjects (8.9%) were categorized in the high-risk with positive FIT group (G1), 173 subjects (18.2%) were categorized in the high-risk with negative FIT group (G2), 192 subjects (20.3%) were categorized in the moderate-risk with positive FIT group (G3), and 499 subjects (52.6%) were categorized in the moderate-risk with negative FIT group (G4). The prevalence of non-advanced and advanced neoplasia in the 4 groups (G1-G4) were 44%/36.9%, 30.1%/11.6%, 27.1%/12% and 22.6%/6.4%, respectively (P<.001) (Figure 1). Subjects in G1 had a significantly higher detection rate of advanced neoplasia (6.15-fold, 95%CI 3.72 to 10.17) than the other 3 groups. Our study demonstrated the synergistic effect of combining FIT with the clinical risk score.
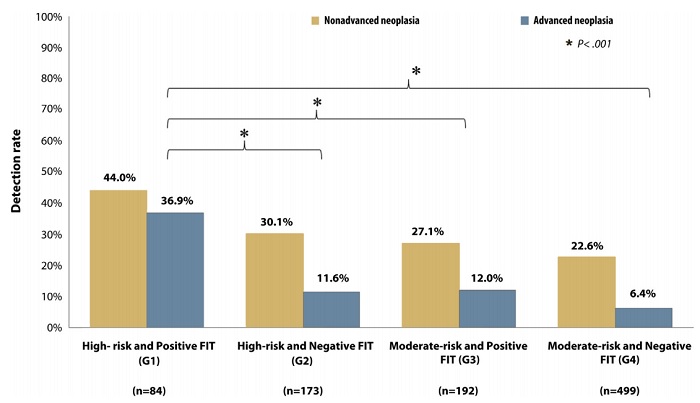
Figure 1. Detection rate of colorectal neoplasia classified by combining clinical risk and FIT stratification. FIT, fecal immunochemical test; G, group.
Because we cannot offer colonoscopy for all with positive stool test in a short period of time, we therefore require a method to stratify these subjects. In this study we found that the clinical risk score performed well enough to help us recruiting those with FIT positive with high risk score to undergo for colonoscopy as the first priority and those with either only high-risk or only FIT positive can undergo for a later colonoscopy. In addition, subjects with FIT negative who has only moderate-risk may undergo colonoscopy as the last group if colonoscopy resource is allowed. However, in countries with very limited resources, a colonoscopy may be omitted in these.
Read the abstract for this article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
