 Kulwinder S. Dua, MD, FASGE, FACP, FRCP from the Department of Gastroenterology and Hepatology at the Medical College of Wisconsin in Milwaukee, Wisconsin, USA reports on his Original Article “Efficacy and safety of a new fully covered self-expandable non-foreshortening metal esophageal stent.”
Kulwinder S. Dua, MD, FASGE, FACP, FRCP from the Department of Gastroenterology and Hepatology at the Medical College of Wisconsin in Milwaukee, Wisconsin, USA reports on his Original Article “Efficacy and safety of a new fully covered self-expandable non-foreshortening metal esophageal stent.”
The primary focus of this study was to evaluate the safety and efficacy of a newly released fully covered, non-foreshortening, self-expandable metal esophageal stent (SEMS).
Periodically, new types of esophageal stents are introduced in the U.S. market and world-wide. The design and characteristics of SEMSs have a bearing on their efficacy and safety profiles. The majority of the esophageal stents being used in the U.S. foreshorten on deployment. Precise positioning of foreshortening stents can be challenging especially when strictures are located near the UES. In these circumstances, a non-foreshortening stent would be preferred. Recently, a non-foreshortening SEMS was released in the U.S. market (EndoMAXX stent, Merit Medical, Utah, Figure 1). It is a fully covered stent and to reduce the risks of migration, it has large anti-migration struts. This stent is coated with silicone that does not degrade as readily as polyurethane. We felt it important to systematically study the performance characteristics of this new stent.
 Figure 1. A new non-foreshortening fully covered expandable esophageal stent. A, The stent comes preloaded onto a 90-cm long, single-hand, delivery system. The maximum diameter of the delivery system is 8 mm. B, Constrained stent. C, Expanded stent. The stent is coated with a silicone membrane internally and externally along its entire length.
Figure 1. A new non-foreshortening fully covered expandable esophageal stent. A, The stent comes preloaded onto a 90-cm long, single-hand, delivery system. The maximum diameter of the delivery system is 8 mm. B, Constrained stent. C, Expanded stent. The stent is coated with a silicone membrane internally and externally along its entire length.
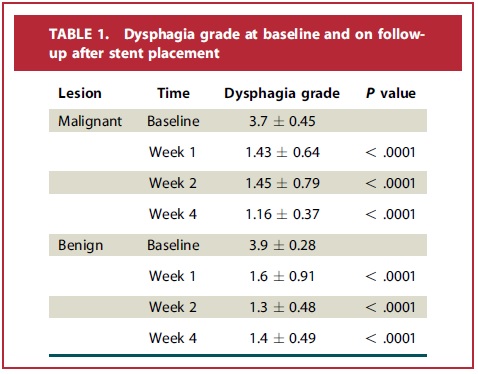
A total of 43 stents were placed in 24 patients with malignant strictures and 11 patients with benign strictures/fistulae/leaks. It was easy to precisely position the stent including in those with strictures within 2 cm of the UES. The technical success in placing and removing (where indicated) the stents was 100%. The stent was effective in relieving dysphagia (Table 1) and sealing /healing benign fistulae and leaks. The majority of the refractory benign esophageal strictures recurred after stent removal. Migration (14%), chest pain (8.5%), and recurrent dysphagia from tissue under-growth (6%) were some of the adverse events noted. Overall, similar to other currently available stents, the recently released SEMS was effective in palliating malignant dysphagia. The non-foreshortening feature of this stent allowed for precise placement and its fully covered feature allowed for removability (not yet FDA cleared for this indication). The stent was also effective in treating benign fistulas and leaks. Adverse events including migration were acceptable.
Compared to the only other non-foreshortening, fully covered stent in the U.S. (Alimaxx-ES, Merit Medical, Utah), the new stent is made of a thicker nitinol tube thereby reducing the possibility of longitudinal in-folding. The silicone membrane on the new stent is less likely to degrade compared to the polyurethane coating of the Alimaxx-ES stent. Unlike the Alimaxx-ES stents, the new stent is not available in smaller diameters (16 mm and 12 mm).
Read the abstract for this article online here.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
