 Charles J. Kahi, MD, MSCR from Indiana University School of Medicine in Indianapolis, Indiana reports on this Original Article “Improving measurement of the adenoma detection rate and adenoma per colonoscopy quality metric: the Indiana University experience.”
Charles J. Kahi, MD, MSCR from Indiana University School of Medicine in Indianapolis, Indiana reports on this Original Article “Improving measurement of the adenoma detection rate and adenoma per colonoscopy quality metric: the Indiana University experience.”
The adenoma detection rate (ADR) is a validated marker of colonoscopy quality. However, there are concerns about its potential for corruptibility, and the optimal methods to measure it.
We reviewed 21,766 colonoscopies and found that the ADR and adenoma per colonoscopy (APC) quality metric were significantly correlated. For men, an ADR of 25% corresponded to an APC of 0.46 (95% confidence interval [CI], 0.35-0.57); for women, an ADR of 15% corresponded to an APC of 0.20 (95% CI, 0.13-0.27). We also found that the ADR stratified by colonoscopy indication was highest for surveillance, followed by screening, then diagnostic. For men, a screening ADR of 25% corresponded to a surveillance ADR of 31.9% (95% CI, 24.8%-38.9%); for women, an ADR of 15% corresponded to a surveillance ADR of 24.3% (95% CI, 18.3%-30.5%). The corresponding diagnostic ADRs were 17.0% (95% CI, 12.4%-21.6%) and 15.4% (95% CI, 11.5%-19.3%), respectively. There was significant correlation between screening ADR and an overall ADR inclusive of all colonoscopy indications. We propose minimum screening APC detection benchmarks of 0.50 for men and 0.20 for women. We also propose that ADRs for screening, surveillance, and diagnostic colonoscopy can be used to derive a simplified overall ADR inclusive of all colonoscopy indications.
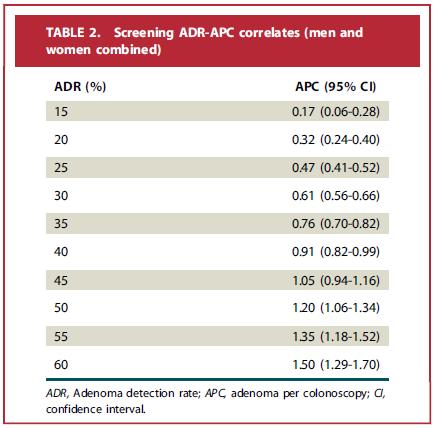
Our study allowed estimation of minimum benchmarks for the APC quality metric, which is less susceptible to corruption and thus a more robust marker of quality than the traditional ADR. The “overall ADR” metric has the potential to simplify ADR calculations because it bypasses the current restriction to screening colonoscopy; this may limit the uptake of the ADR because the process of deriving and calculating the metric for a subgroup of patients may be perceived as labor intensive.
Read the abstract of this study here.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
