Post written by Mark Hanscom, MD, from Weill Cornell Medicine, New York, New York, USA.

This document from the American Society for Gastrointestinal Endoscopy Technology Committee is a comprehensive review of endoscopic devices available for closing transmural defects within the GI tract. Three distinct transmural defects discussed in this document include perforations, leaks, and fistulas, for which the optimal management of each depends on several factors including etiology, acuity of injury, defect size, location, and clinical presentation.
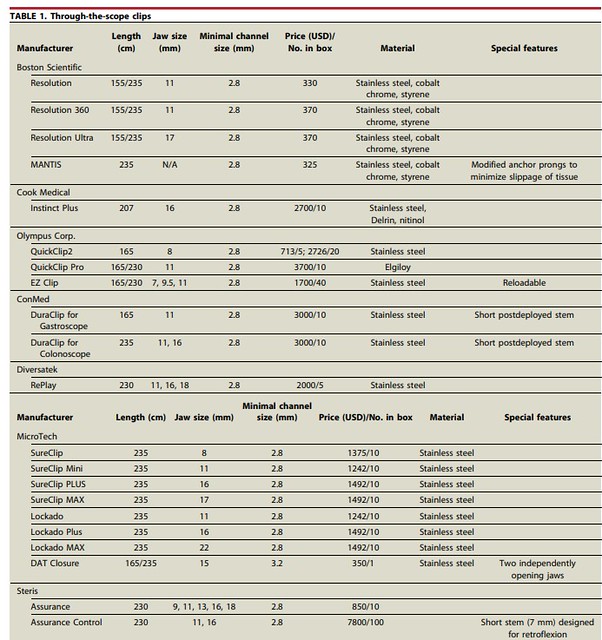
Endoscopic closure is now often considered for the initial treatment of these defects and, as such, the number of endoscopic modalities for treatment has rapidly proliferated. We review the spectrum of technologies for endoscopic closure including endoscopic clipping (through-the-scope [TTS] and over-the-scope clips), over-the-scope suturing, TTS suturing, and application of tissue sealants. For each device, we sought to describe the technical aspects of its use, clinical evidence for its efficacy (where available), and safety considerations.
The landscape of endoscopic closure has changed dramatically over the past decade. Since the previous iteration of this document1 was published, newer endoscopic clip designs–such as the anchor-pronged MANTIS endoscopic clip (Boston Scientific, Marlborough, Mass, USA) and Dual Action Tissue Closure Device (Micro-Tech Endoscopy, USA Inc, Ann Arbor, Mich, USA)–have expanded the capabilities of TTS closure.
In addition, endoscopic suturing platforms have become widespread and introduced a new means of achieving durable endoscopic closure of even large defects not thought amenable to traditional endoscopic clip—based closure. At the same time, the volume and complexity of therapeutic endoscopic procedures, such as endoscopic submucosal dissection, have increased, turning defect closure into a more routine clinical challenge. A consolidated, up-to-date review covering the breadth of these technologies was clearly needed.
Endoscopic closure of GI defects is a common clinical challenge all endoscopists encounter. Whether facing a postpolypectomy perforation or chronic gastrostomy-tube associated gastrocutaneous fistula, all endoscopists will come up against a leak, perforation, or fistula at some point during their career. With advancements in endoscopic closure technologies, most GI defects can now be closed endoscopically, saving patients from the need for emergency surgery. Recognition of and familiarity with the vast array of endoscopic closure devices now available, as well as their application throughout the GI tract, are critical for achieving successful closure and optimal patient outcomes.
This document provides endoscopists with a practical framework for device selection based on defect type, size, location, and chronicity. Technical tips, side-by-side device comparisons, and accompanying instructional videos will hopefully make this a resource that can be used during endoscopy training and at the point of care alike.
One overarching message from this document is that no single device closes all defects optimally. Successful endoscopic closure often requires a multimodal, sequential approach tailored to the individual patient and defect. We hope this review encourages endoscopists to become familiar with multiple closure platforms and to engage in multidisciplinary discussions with surgery and interventional radiology when needed.
Read the full article online.
The information presented in Endoscopedia reflects the opinions of the authors and does not represent the position of the American Society for Gastrointestinal Endoscopy (ASGE). ASGE expressly disclaims any warranties or guarantees, expressed or implied, and is not liable for damages of any kind in connection with the material, information, or procedures set forth.
- ASGE Technology Committee; Banerjee S, Barth BA, Bhat YM, et al. Endoscopic closure devices. Gastrointest Endosc 2012; 76:244-51. ↩︎
